PUTTING ACCESS
INTO PRACTICE
MEDICAL ABORTION ON YOUR TERMS
Medical Abortion & Mifepristone
In the US, an increasing number of people are choosing medical abortion to end an early pregnancy. Medical abortion is an FDA-approved method to end a pregnancy within the first 70 days after a patient’s last menstrual period. It involves taking two medications: mifepristone, which blocks a hormone needed for a pregnancy to continue, and misoprostol, which causes contractions to expel the pregnancy. Mifepristone is available only under an FDA-administered Risk Evaluation and Mitigation Strategy (REMS) program. Until 2019, when GenBioPro launched its generic medication, mifepristone was only available in the US as a branded product. The introduction of GenBioPro generic mifepristone tablets, 200 mg, has expanded access to medical abortion through stabilized product supply, improved packaging, seamless ordering, and timely fulfillment.
Medical abortion has been used safely and effectively in the US for more than 20 years. Earlier changes to the mifepristone label and REMS guidelines expanded who may prescribe generic mifepristone tablets, 200 mg, as well as the window of time it may be used by patients. Other changes in 2023 have further increased access:
- Removing the requirement for in-person dispensing, allowing telemedicine care in states that permit it.
- Allowing mail and in-person dispensing from certified pharmacies.
- Streamlining the Prescriber Agreement so that it can be used wherever a health care provider practices and prescribes.
- Clarifying the REMS program guidelines and updating the full prescribing information and the Medication Guide.
FDA-Approved Generic Mifepristone Tablets, 200 mg, from GenBioPro
Generic mifepristone tablets, 200 mg, from GenBioPro, the first FDA-approved generic medical abortion pill in the US, are now available for certified pharmacies to stock and dispense upon receipt of a prescription from a certified prescriber.
The FDA determined that GenBioPro generic mifepristone tablets, 200 mg, are equivalent to the branded drug.
Certified pharmacies can benefit from being better able to serve customers, expanded product access, confidence in a stable product supply, and pharmacy-friendly packaging options with seamless ordering and timely fulfillment for generic mifepristone tablets, 200 mg, from GenBioPro.
Dispensing Regulations
Mifepristone is available only under an FDA-mandated Risk Evaluation and Mitigation Strategy (REMS) program. To become a certified pharmacy and fill prescriptions for GenBioPro mifepristone tablets, 200 mg, a pharmacy must be able to meet the requirements of the REMS program. For example, certified pharmacies must receive and retain a completed and signed Prescriber Agreement from any prescriber for whom they fill prescriptions. See detailed REMS requirements.
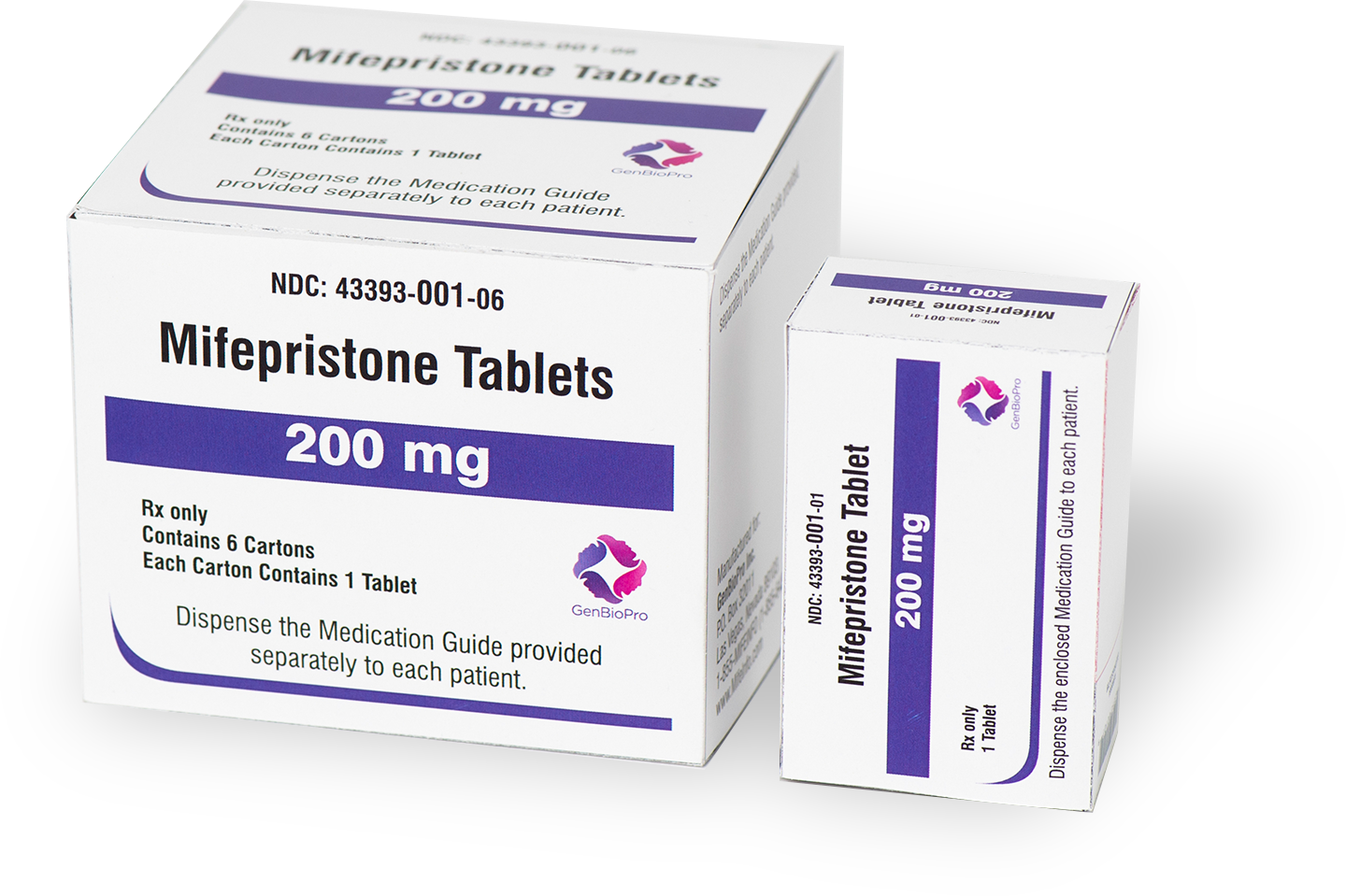
Product Specifics
GenBioPro generic mifepristone tablets, 200 mg, are FDA-approved and equivalent to the branded product for medical abortion up to 70 days gestation.
- AB rated to: Mifeprex®
- ANDA Number: 091178
- Date of FDA Approval: 04/11/2019
- Ingredients: A light yellow tablet containing 200 mg of mifepristone. Inactive ingredients include colloidal silicon dioxide, corn starch, povidone, microcrystalline cellulose and magnesium stearate.
- Storage Information: Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F). Protect from light.
- Tablet Description: The tablet is light yellow in color, circular, biconvex, and is intended for oral administration only
- Additional Information: For adverse event and product quality complaints, please call 1-855-MIFEINFO (1-855-643-3463).
Need More Information?
If you have questions about GenBioPro generic mifepristone tablets, 200 mg, becoming a GenBioPro certified pharmacy, or ordering product, contact GenBioPro at:
Online | 1-855-MIFE-INFO (1-855-643-3463) | info@genbiopro.com
